Variety of languages: German, English, French, Spanish.Economical handling of access rights on the smartpohne app.Offline use. No Internet needed - applies only to the smartphone app.Elemental comparison - compare elements values right next to each other.The web app features a completely overhauled molar mass caluclator.Molar mass calculator: Simple entry field for chemical formulas.Visualized element properties: Atomic radius, atomic radius graphic, electronegativity (according to Allred-Rochow and Pauling), ionization energy, relative atomic mass, state of matter, ranking list of properties, discovery, classifications.All important information about the elements: Atomic number, valence electrons, oxidation state, electronegativity according to Allred-Rochow and Pauling, atomic mass, boiling point, melting point, atomic radius, density, history, discoverer, classification, crystalline structure type, electron configuration, basic state, ionization energy, isotopic composition, state of matter, hardness according to Mohs, oxidation numbers, percentage of mass in Earth's crust, year of discovery, half-life, and much more.Access the same amount of information on your browser, faster and larger than ever before with the same renowned ease of use.ĭownload now, or access directly online, free of charge and start experimenting. Our smartphone app (with over 4.89 million downloads!) is now joined by our newest update which features an online web version of our famous PTE smartphone app. Get informed with our mobile reference work, any time, with ease, offline (smartphone app) and in detail. Our app is a must-have of digital periodic tables. Hint: Count the number of atoms of each element, and then multiply that number by the element's atomic weight.The Merck KGaA, Darmstadt, Germany PTE App is the ultimate tool for every friend of chemistry - whether pupil or teacher, student or professor, amateur or expert, hobbyist or technician.
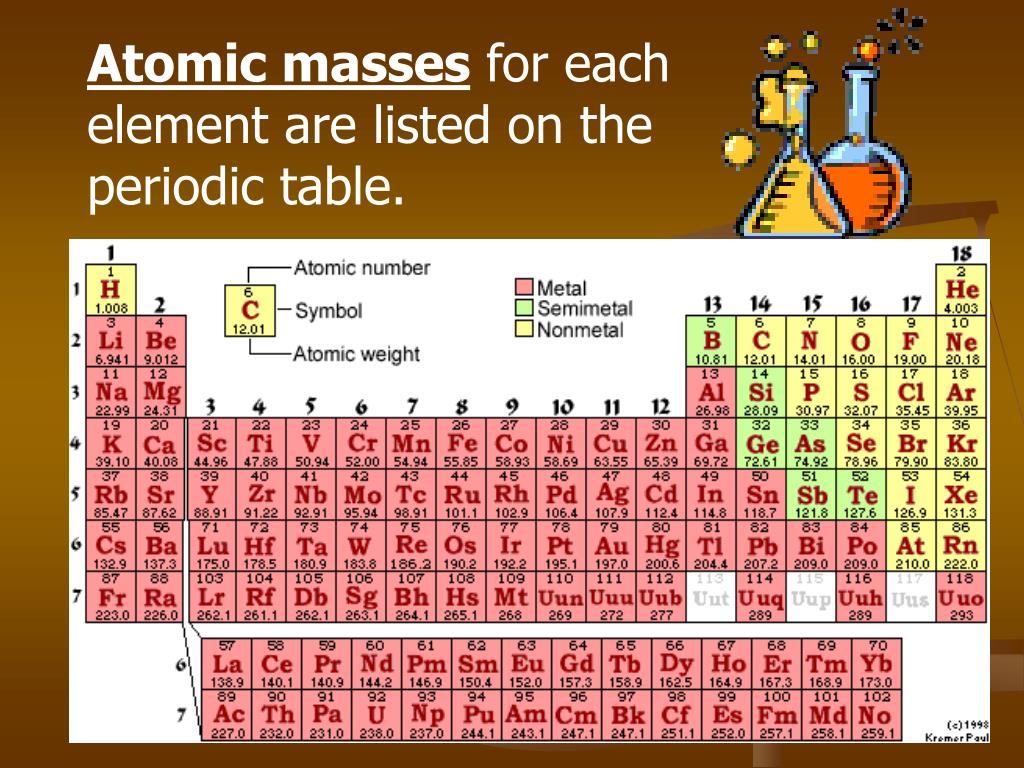
Use the table below to find the atomic weight of each atom (element), or refer to a Periodic Table of the Elements. Let us calculate the molecular weight of some common compounds. For example, in one mole of a chemical compound there are 6.022 x 1023 molecules. One mole of 'something' contains 6.022 x 1023 entities. A mole is the unit that measures the amount of a substance. One thousand mers connected together would add up to a weight of 28,000 grams/mole and would have 6,000 atoms.Ī mole is the standard method in chemistry for communicating how much of a substance is present. We combine (react) many mers of ethylene together to form a polyethylene chain. The atomic weight of carbon is 12 and that of hydrogen is 1, so one mer of ethylene has a weight of 2(12) + 4(1) = 28. It has a total of 6 atoms: 2 carbon (C) atoms and 4 hydrogen (H) atoms.

The chemical formula for an ethylene monomer is -(CH2-CH2).In order to calculate the molecular weight of one water molecule, we add the contributions from each atom that is, 2(1) + 1(16) = 18 grams/mole.

Using the periodic table of the elements to find atomic weights, we find that hydrogen has an atomic weight of 1, and oxygen's is 16. The chemical formula for water is H2O, which means this molecule has 3 atoms: 2 of hydrogen (H) and 1 oxygen (O) atom.
