
Lead's high density, low melting point, ductility and relative inertness to oxidation make it useful. In 2014, the annual global production of lead was about ten million tonnes, over half of which was from recycling. Lead played a crucial role in the development of the printing press, as movable type could be relatively easily cast from lead alloys. Lead production declined after the fall of Rome and did not reach comparable levels until the Industrial Revolution.
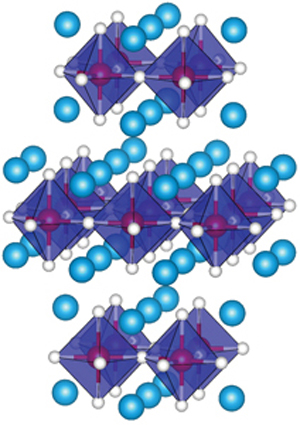
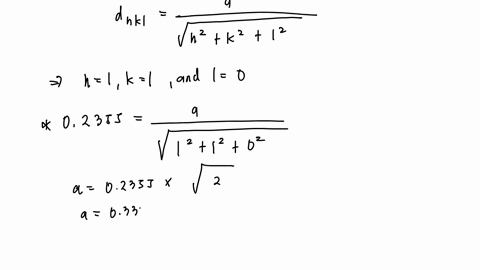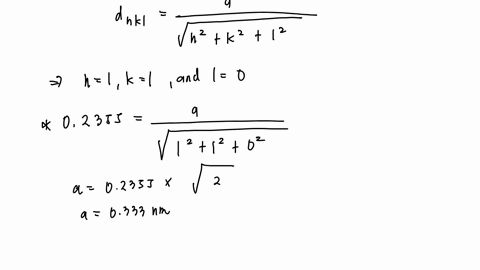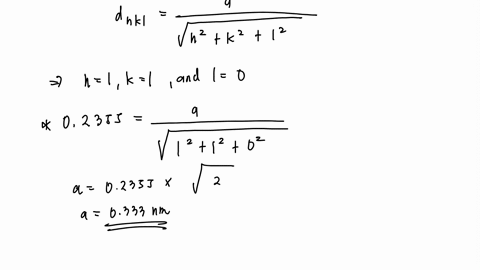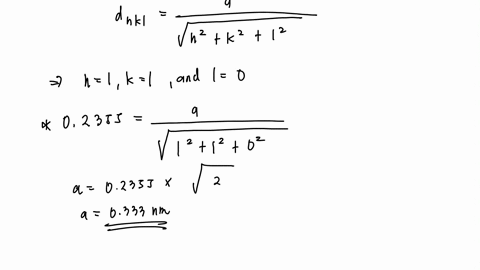
Interest in silver helped initiate widespread extraction and use of lead in ancient Rome. Galena is a principal ore of lead which often bears silver. Since lead is easily extracted from its ores, prehistoric people in the Near East were aware of it. Like the lighter members of the group, lead tends to bond with itself it can form chains and polyhedral structures. Exceptions are mostly limited to organolead compounds. Compounds of lead are usually found in the +2 oxidation state rather than the +4 state common with lighter members of the carbon group. Its weak metallic character is illustrated by its amphoteric nature lead and lead oxides react with acids and bases, and it tends to form covalent bonds. Lead is a relatively unreactive post-transition metal. Lead is toxic, even in small amounts, especially to children. Lead has the highest atomic number of any stable element and three of its isotopes are endpoints of major nuclear decay chains of heavier elements. It tarnishes to a dull gray color when exposed to air. When freshly cut, lead is a shiny gray with a hint of blue. Lead is soft and malleable, and also has a relatively low melting point. It is a heavy metal that is denser than most common materials. At some specific conditions, silver can also show HCP (hexagonal close packing) structure.Lead is a chemical element it has symbol Pb (from Latin plumbum) and atomic number 82. Always remember, the noble metals (copper, silver, gold) crystallise in fcc structure at room temperature. Note: Metals that have fcc structure are gold, copper, iridium, lead, nickel, aluminum, platinum and silver. Therefore, we can conclude that silver metal has FCC type crystal structure. Low hardness and high ductility of silver metal is due to the fact that unlike other metals with incomplete d-shells, it has metallic bonds with low covalent character and are relatively weak.
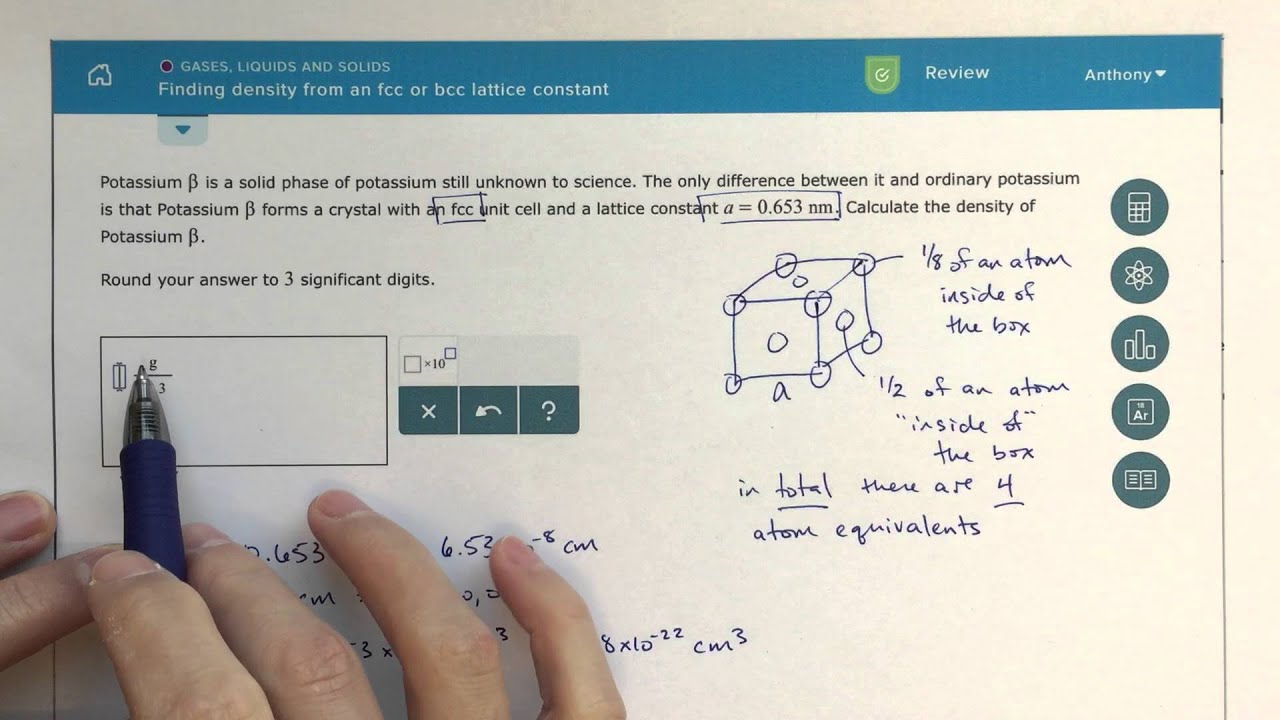
Silver is an extremely soft, ductile and malleable metal. Fcc is also known as cubic close packing(ccp). It has coordination number $12$ (total no of nearest neighbouring central atom is having in the surrounding). Fcc has a cubic structure with an atom on each face and corner of the cube. As we know that, fcc has a packing sequence of ABCABC, such that rows of atoms in C layer are arranged in such a manner that they do not overlap atoms in either A or B layer, it is the closest possible packing of spheres leaving about only one fourth of total space available. Silver metal has a crystal structure of face centered cubic (fcc) lattice with bulk coordination number $12$ where only $5$ Central atom has a coordination number of $12$. The crystal structure of silver metal consists of a total four atoms in a unit cell i.e., eight eighth of atoms at corners and halves of atoms at six faces (cube has eight corners and six faces). Hint: The structure of metal solid is made up of metal ions arranged in a closely packed manner.
